Electro Oxidation Basics#5: Why Choose BDD Electrode Over lead dioxide electrode
BDD Electrode Vs. Lead Dioxide Electrode, A Showdown And Engineering Guide To High Performance Oxidation
Welcome to the 5th content of electro oxidation wastewater treatment basics, and this context is about another electrode material, lead dioxide electrode, as one of the critical anode for electrochemical oxidation wastewater treatment processes which is one of the Advanced Oxidation Processes (AOPs), lead dioxide anode performed well when economic issues be the major concerns since it does not require expensive substrates, then boosting mass transfer and offer high oxidation kinetics, as this group of anodes can easily applied to 3D substrates (e.g., Reticulated Vitreous Carbon – RVC/), and suitable for large-scale, low-cost applications, improved stability, capability and efficiency to remove COD, total organic carbon (TOC), and specific organic pollutants such as RNO, and certain groups of pesticides.
Since this content is all about a showdown of Boron-Doped Diamond BDD electrode and lead dioxide electrode. These types of electrode materials are mainstream anode in electro oxidation wastewater treatment processes to degrade and mineralize organic compounds, and important components of industrial wastewater treatment solutions, therefore this context will cover physci-chemical properties, differences with mechanisms in wastewater treatment and organic pollutants removal, side-by-side comparison of electrochemical efficiency, electrode lifespan, degradation efficiency toward general organics and persistent organic pollutants, followed by applications, total cost of ownship, conclude with a comparative comparison of these two electrodes.
Content regarding lead dioxide is inspired by application of lead oxide electrodes in wastewater treatment from sciencedirect.
- Data And Information From Our Team
More About Electro Oxidation
Searching for your own types of anode material or wastewater treatment approach to degrade organic pollutants from all these complex industrial wastewater?
Search no more, simply ask our engineering team how boron doped diamond electrode can tackle all the challenges you are facing now.
A Showdown: BDD Electrode vs. Lead Dioxide Electrode
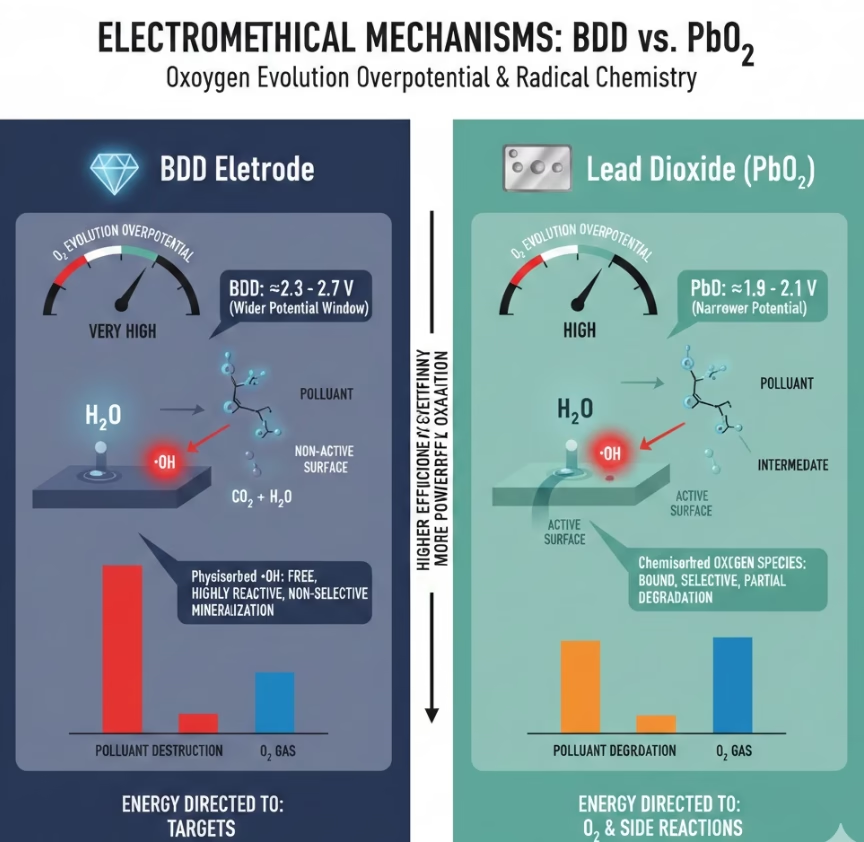
BDD Electrode Vs. Lead Dioxide Electrode On Oxygen Evolution Overpotential
The Oxygen Evolution Overpotential (OEP) is one of the most critical metrics for an Electrochemical Advanced Oxidation Processes (EAOPs) when it comes to selecting electrode materials, especially anode, OEP is the voltage at which water begins to electrolyze into oxygen gas (O2).
Lead Dioxide (PbO2) has a high OEP at approximately 1.9V. Therefore it can be effective at reactive oxidant generation, however a critical percentage energy is divided to oxygen generations.
Borond doped diamond BDD electrode on the other hand, with an OEP at some 2.3v to 2.7v which is the widest potential window of any known electrode material available for industrial applications. This allows the electrode to reach much higher energy levels before water interference occurs, directing more energy toward the destruction of persistent organic pollutants.
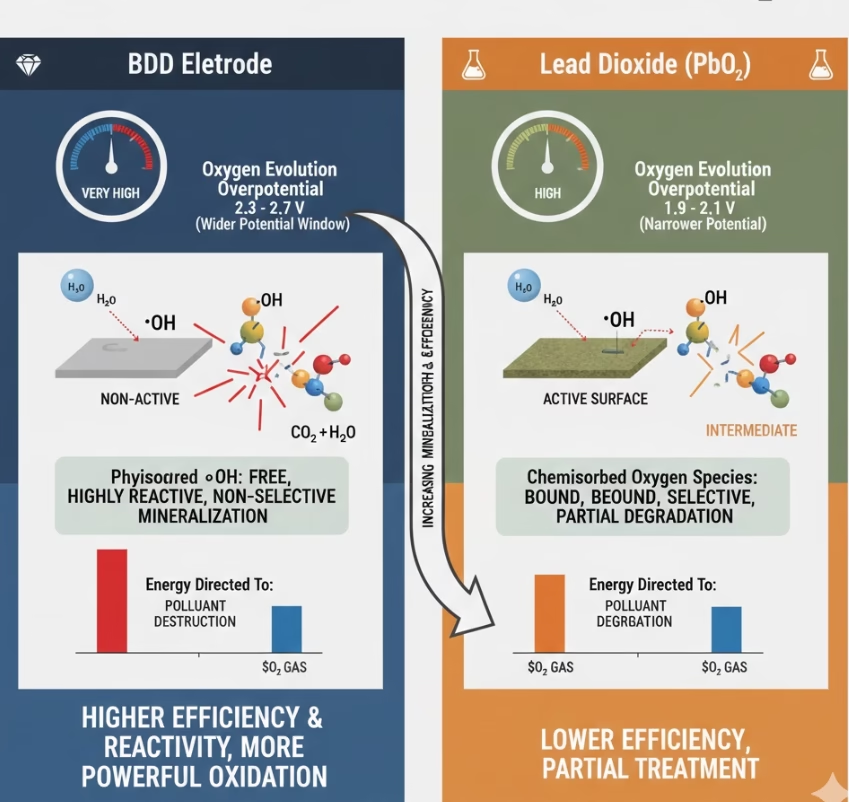
Lead Dioxide Electrode Vs. BDD on Depth of Oxidation & Mineralization
As an “active” anode, reactive oxygen oxidants such as hydroxyl radicals , ozone, and other oxidizing agents are strongly adsorbded to the surface of the lead dioxide electrode while it reacts with water, hydroxyl radicals strongly adsorb to the anode surface, forming higher-state metal oxides, that is transforming PbO2 into PbO3. Therefore It is excellent for selective oxidation, for instance, converting an alcohol to an aldehyde, however it can not break chemical bonds of peristent organic pollutants, leaving intermediates and POPs intact.
Reactive radicals are physisorbed on the surface of diamond electrode as the inert properties of the non-active BDD electrode eventually form thin layers of hydroxyl radicals insteading trapping them like lead dioxide anodes—they float in a thin layer just above the surface, highly mobile and extremely aggressive. These hydroxyl radicals are hight reactive and mobile, they interact with all types of organic molecules, break chemical bonds non-selective way, eventually degrade most of the pollutants.
Lead Dioxide Electrode Vs. BDD Electrode for Wastewater Treatment
This next section is a comprehensive comparison of lead dioxide electrode and boron doped diamond electrode on different aspects of wastewater treatment, mainly chemical oxygen demand (COD), total organic carbon (TOC), persistent organic pollutants (POPs) removal, and treatment efficiency of these two electrodes toward target pollutants, followed by a comparative comparison with a radar chart, which electrode to choose when it comes to different water matrixs, cost of ownship, stability and durability in specific application of wastewater treatment.
The efficiency of general COD degradation, comprehensive TOC removal, and persistent organic pollutants follows different mathematical paths when it comes to these two anode materials, therefore we create the following part as a kinetic comparison of lead dioxide electrode and boron doped diamond electrode, we hope this specific content will help you to get a better comprehension about differences of these electrodes, that is the efficiency gap, especially after we discussed the oxidation and mineralization part within the section above.
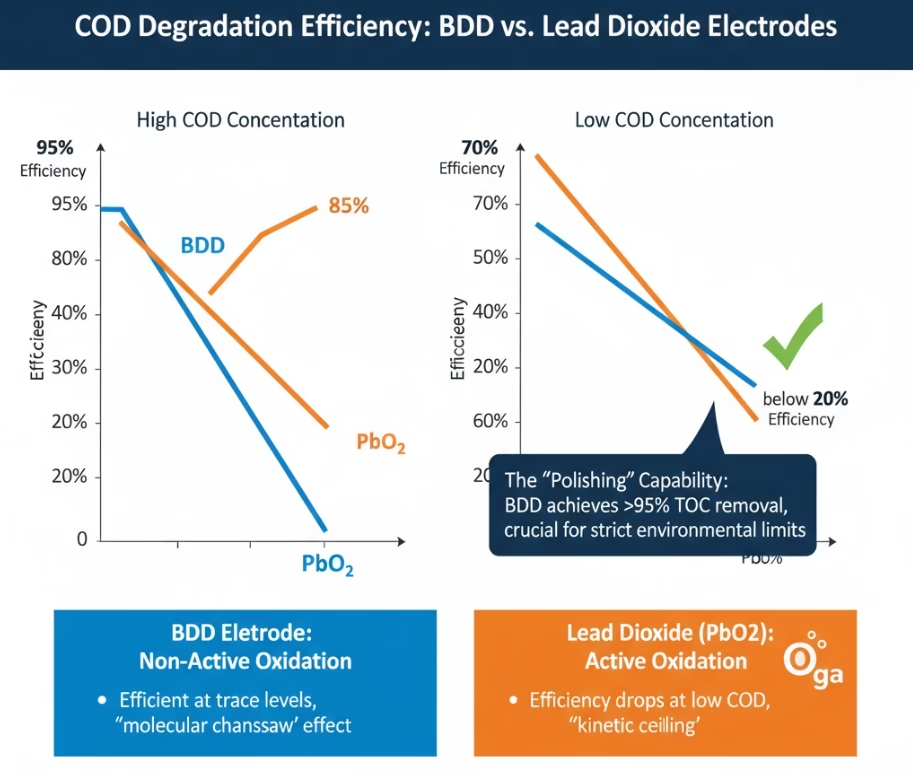
COD Degradation Efficiency Comparison of BDD vs. Lead Dioxide Electrode
The COD degradation with lead dioxide is always restrained by the “active” nature of the PbO2 surface. Organic molecules need to be physically adsorbed onto the lead sites to be oxidized, which generate blind spots on the surface, especially if the wastewater has “fouling” agents like proteins or fats. Therefore lead dioxide anode emits a slower, linear COD degradation rate that struggles to reach untraceable level.
Organic pollutants mineralization COD degradation rate of BDD anode reaches some 92%, yet save 30-55% of the time with PbO2 anode when it comes to complex industrial waste streams, as the whole surrounding area of the “non-active” BDD anode is a highly intensive oxidation zone. What is more, BDD is the only anode with a polishing capability of slashing COD from 500 mg/L down to <10 mg/L in a single process.
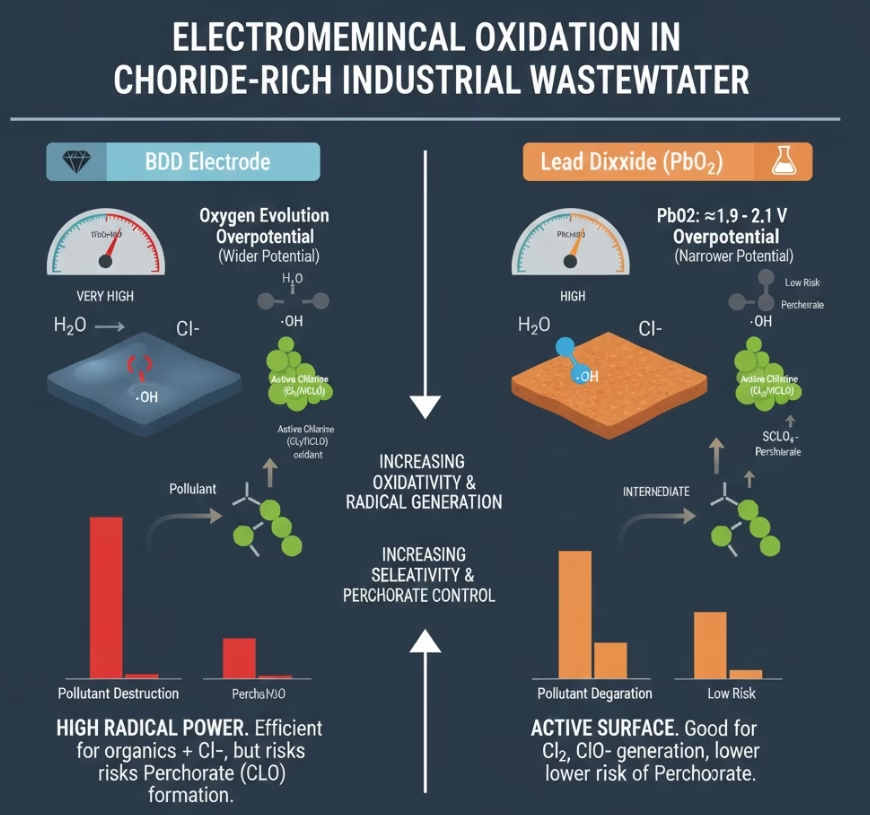
Anode Selection for Different Water Matrix #1: Chlorides & Active Chlorine
Industrial wastewaters such as chemical & petrochemical, textile, then pharmaceutical systhesis, produced water from oil and gas, food processing, leather tanning, and etc, are with high concentration of chlorides.
In a high-chloride waste streams, radical machine like BDD anode can generates active chlorine species such as Cl2, HClO, ClO-, as secondary oxidants besides active oxygen species. However, BDD electrode can oxidate chlorides into perchlorates ClO4-, which is a harmful byproduct.
PbO2 lead dioxide is a better choice for chloride-rich environments. It has less chance to generate significant perchlorate levels compared to BDD under certain conditions, making it a “safer” choice when discharge permit has strict limits on perchlorate but allows for active chlorine.

Anode Selection for Different Water Matrixs #2: Sulfate Rich Waste Stream
Sulfate rich conent are common in wastewater generated from mining, mineral processing, chemical and manufacturing sectors, pulp and paper, food and beverage processing.
Let’s check which one to choose for sulfate rich waste streams.
Sulfates are oxidized into persulfates (S2O82-) on the surface of the BDD anode. Which put the “extra” indirect oxidation via persulfates to the in existing direct oxidation via hydroxyl radicals, indirect oxidation on the anode surface in this scenario. In this case, BDD electrode can reach a much higher redox kinetic rates for organic pollutants removal, thus boost treatment efficiency.
While PbO2 lead dioxide electrode can handle sulfates, however, it can not transfer sulfates into persulfates with the same efficiency like BDD.
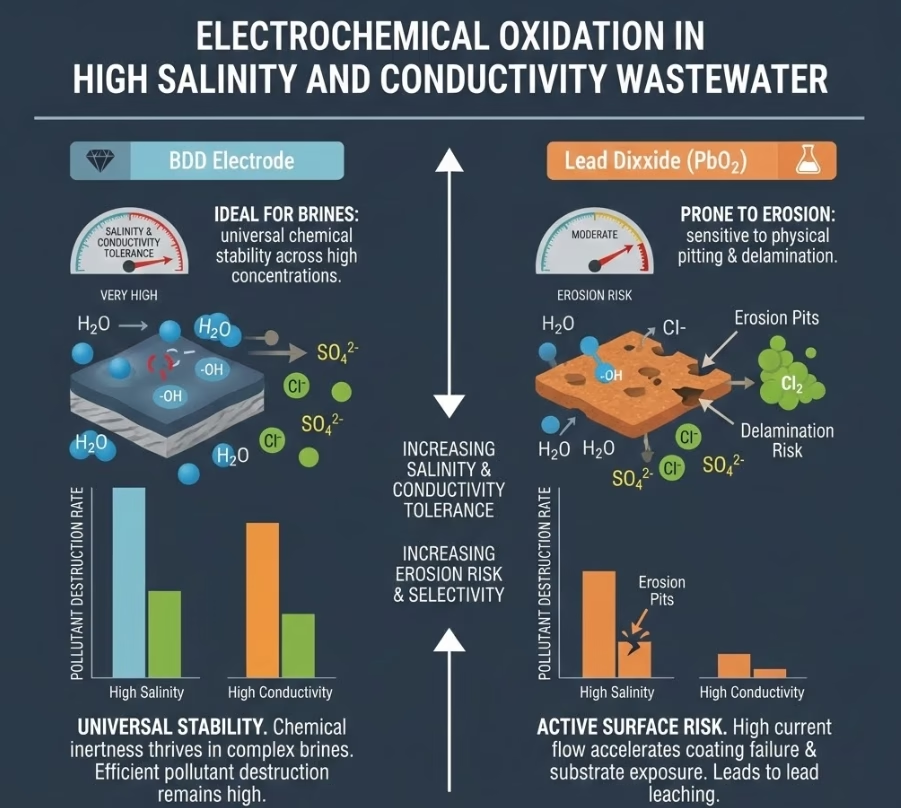
Anode Selection for Water Matrixs #3: High Salinity and Conductivity
Wastewater originated from tannery, pharmaceuticals, petrochemical and refining, power plants, then produced water from oil and gas, and etc, are with high salinity, therefore highly conductive, and usually with high loads of organic pollutants.
In high-salinity water, PbO2 coatings of the lead dioxide electrode are susceptible to enhanced physical erosion. The aggressive evolution of gas at the surface can cause “pitting” in lead dioxide layer, eventually exposing the titanium substrate to corrosion,therefore PbO2 anodes demands high maintenance and frequent replacement.
BDD anode, on the hand, is essentially immune to salinity-induced corrosion. It thrives in high-conductivity environments, allowing you to run at higher current densities without fear of the diamond film delaminating or the substrate failure.
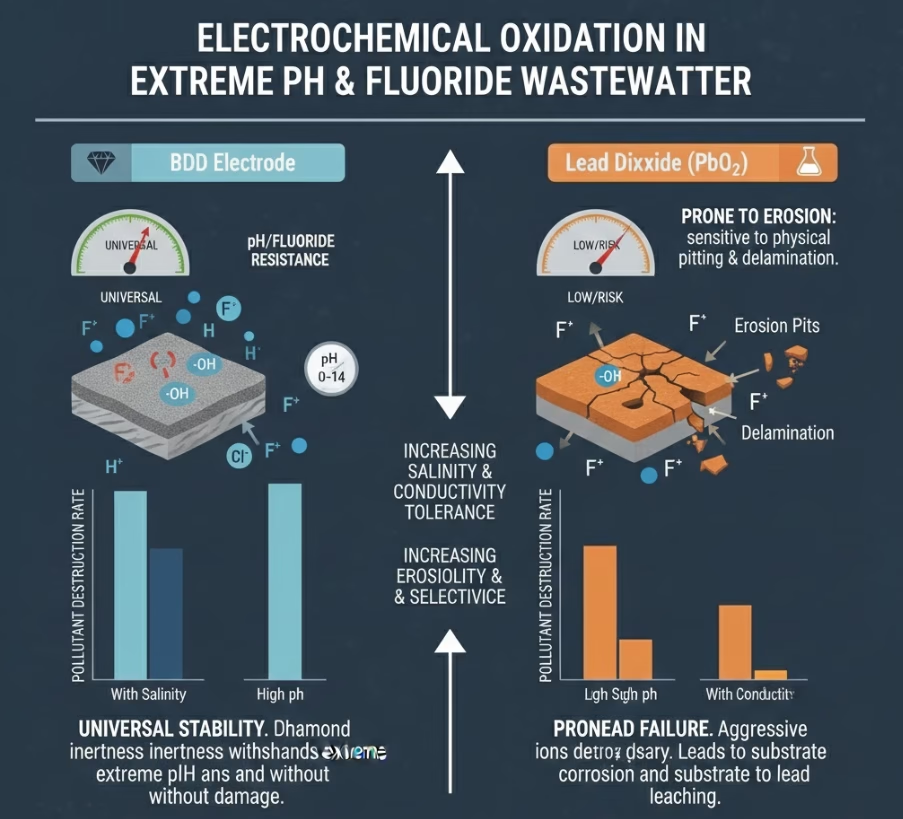
Anode Selection for Different Water Matrixs #4: Extreme pH and Fluorides
Wastewater from semiconductor, metallurgy, electroplating, metal smelters, photovoltaic, electronics, Li-ion battery manufacturing & recycling sectors are with high concentrations of fluorides.
Fluorides aggressively attack the titanium/lead-oxide interface, causing the coating to peel off in weeks. BDD electrode grown on niobium or silicon substrates is one of the few materials that can survive in hydrofluoric acid (HF) environments.
Industrial waste streams from textile,metal finishing, spent caustic, pharmaceuticals, chemical productions, semiconductor and etc, are with wide pH fluctuations, PbO2 performs best in acidic to neutral conditions, while unstable in strongly alkaline matrices. BDD anode is chemically stable across the entire pH scale (0–14).
Questions about electrode material? Get started to reaching out to us now.
It's strategic decision balancing upfront investment against long-term operational performance and environmental compliance of different electrode materials.