Electro Oxidation funamentals: Science Behind Electrochemical Advanced Oxidation processes Explained
The Science of Electrochemical Advanced Oxidation Processes (EAOPs)
As one of the most innovative and most efficient advanced oxidation proceses (AOPs), Electrochemical Advanced Oxidation Processes, EAOPs for short, established a trackable efficient record toward waste streams, especially industrial wastewater treatment with electrochemical combustion, oxidation then mineralization of persistent organic pollutants (POPs) into carbon dixoide, water, and inorganic salts, instead of breaking water molecules into offering a robust solution for the degradation of persistent organic pollutants (POPs) that traditional biological or chemical treatments fail to address. At the heart of this technology is the “Anodic Oxidation” process, where electrical energy is harnessed to generate highly reactive species directly at the electrode surface.
Unlike standard electrolysis, which might focus on simple redox reactions, EAOPs are engineered to achieve mineralization—the complete conversion of complex organic molecules into water, carbon dioxide, and inorganic salts.
Active vs. Non-Active Anodes: The Differences with Oxidation Mechanism
The efficiency of an EAOP wastewater treatment system is primarily dictated by the properties of the anodic electrode material. Anode electrode materials function as electrocatalytic function, or say catalyst electrode material types are devided into two paralleled categories, active anode and non-active anode, based on how each group of anodes interact with the major oxidant in electrochemical advanced oxidation process, hydroxyl radicals (ᐧOH), feel free to check out how hydroxyl radicals functions in wastewater treatment via the hydroxyl radical for wastewater treatment content.
Active Anodes And Hydroxyl Radical Generation
Actice anodes generate hydroxyl radicals via the pathway within the right section: MOx + H2O ⟶ MOx(ᐧOH) + H⁺+ e⁻
Active Anodes such as IrO₂, RuO₂, Pt interact strongly with ᐧOH, often leading to a high rate of further oxidation to higher-valence metal oxides (MO𝑥), which slashes the amount of ᐧOH available for water treatment, which means a lower current efficiency for organic degradation, followed by a comparatively much lower organic pollutants removal efficiency.
These anodes typically favor selective oxidation or electrochemical conversion. Because the radicals are “trapped” on the surface, they are less available for the unselective destruction of complex organics, often leading to partial degradation, that is intermediates generation instead of further oxidation, then full mineralization.
Non-Active Anodes And Hydroxyl Radical Generation
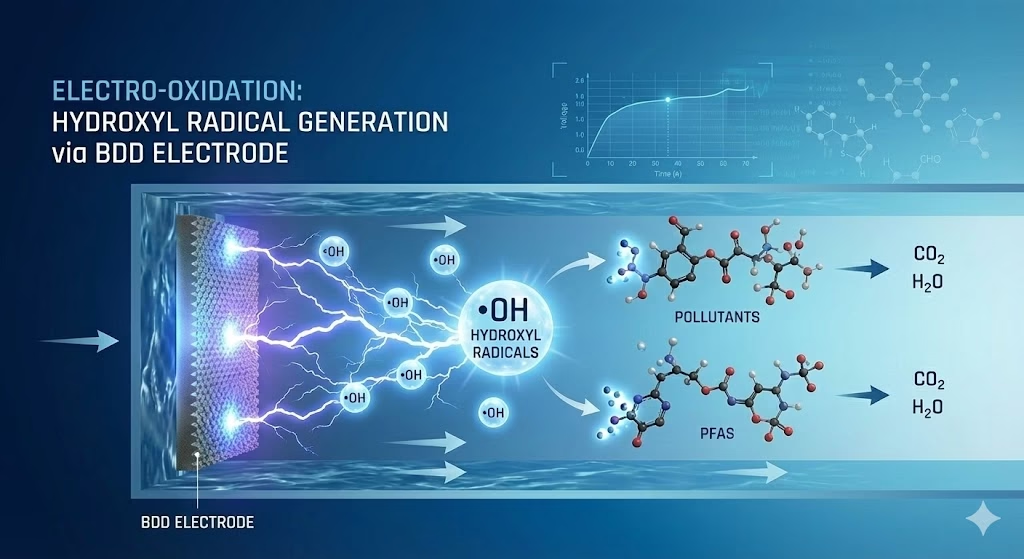
Non-Active anodes exhibit “inert” surface properties with very low adsorption energy for hydroxyl radicals. This allows the radicals to remain “physically adsorbed” and highly mobile.
Hydroxyl radicals generation via non-active anodes follow this pattern: M + H2O ⟶ M(add ᐧOH) + H⁺+ e⁻ , these non-active anodes facilitate unselective oxidation. The ᐧOH radicals exist in a quasi-free state, possessing a much higher oxidation potential. This is the preferred mechanism for organic compounds oxidation, then enhance possibility of complete mineralization, as the hydroxyl radicals are aggressive enough to break down even the most stable carbon-fluorine or aromatic bonds by non-selective oxidation via their four unique pathways: hydrogen abstraction, addition to unsaturated bonds, electron transfer, then radical combination.
Hydroxyl Radical (ᐧOH) Life Cycles in Aqueous Solutions
With an electrochemical oxidation potential at 2.8v, a much higher oxidation potential than other reactive oxygen species, hydroxyl radicals can conduct non-selective oxidation toward persistent organic pollutants into intermediates, aka simpler compounds ready for further oxidation, while ozone and other ROS disinfect, these advantages make hydroxyl radicals the “workhorse” of EAOPs.
Understanding its lifecycle—from generation to scavenging—is critical for optimizing electrochemical reactor power consumption and flow rates.
Hydroxyl Radical (ᐧOH) Generation Phase
Hydroxyl radicals are produced via the oxidation of water molecules at the anode surface once the potential exceeds the thermodynamic stability of water. This requires an anode with a high Oxygen Evolution Overpotential (OEP). If the OEP is too low, the energy is wasted on producing oxygen gas (O₂) rather than (ᐧOH).
Hydroxyl Radical (ᐧOH) Propagation and Reaction
The lifetime of a hydroxyl radical is incredibly short—measured in nanoseconds. It must react almost instantly with a target pollutant (R) via:
Hydrogen Abstraction: Removing H atoms from saturated compounds.
Electrophilic Addition: Attacking unsaturated double bonds or aromatic rings.
Electron Transfer: Direct oxidation of functional groups.
Hydroxyl Radical (ᐧOH) Termination and Scavenging
If the radical does not encounter a pollutant, it enters a termination phase. It may recombine to form hydrogen peroxide (H₂O₂) or be “scavenged” by bicarbonate or chloride ions present in the water. Effective EAOP design ensures that mass transfer rates (the movement of pollutants to the electrode) match the radical generation rate to minimize these “wasted” cycles.
Boron-Doping in sp³/sp² Ratios On Boron Doped Diamond BDD Electrode
Boron-Doped Diamond (BDD) electrode is widely considered the “gold standard” for non-active anodes.
However, its performance is not universal; it is determined by the atomic structure of the diamond film during the Chemical Vapor Deposition (CVD) process.
The sp³ / sp² Balance with BDD Electrode, Why It Matters
sp³ Carbon: This represents the tetrahedral diamond lattice. A high sp³ content provides the chemical inertness, corrosion resistance, and the wide potential window (high OEP) necessary for efficient hydroxyl radicals production.
sp² Carbon: This represents “graphitic” impurities. While some sp² carbon can increase conductivity, an excess reduces the OEP. Too much graphitic content causes the anode to behave more like an “active” electrode, leading to surface fouling and lower oxidation efficiency.
Boron-Doping Levels with Borond Doped Diamond (BDD) Electrode
The introduction of Boron atoms into the lattice transforms the diamond from an insulator into a semiconductor.
Low Doping: Results in high resistance and poor current distribution.
High Doping (>2000 ppm): Leads to “metallic” conductivity.The engineering challenge lies in achieving a high Boron concentration (for conductivity) without collapsing the sp³ diamond structure into sp² graphite.
By maintaining a high sp³/sp² ratio, BDD anodes can operate at high current densities without degrading, providing the most stable environment for long-term industrial wastewater remediation.
- Ideal Anode Material for Electrochemical Advanced Oxidation Processes for you
Premium Quality Borond Dopd Diamond BDD Electrode
By integrating these fundamentals—matching the specific anode mechanism to the pollutant profile and optimizing the sp³ structural integrity, engineers can design EAOP systems that significantly outperform traditional chemical oxidation in both longevity and environmental impact.
Find the right anode with perfectly balanced sp³ / sp², boron doping level, ideal material for your electrochemical advanced oxidation processes applications, click a take closer look now